What is the correct order of reactivity of alcohols in the following reaction?
R-OH + HCl \( \xrightarrow[]{ZnCl_{2}}\) R-Cl + H2O
1. 1° > 2° > 3°
2. 1° < 2° > 3°
3. 3° > 2° > 1°
4. 3° > 1°> 2°
Match item in colum I with those in column II.
| Column I | Column II |
a.  |
i. 2-Ethoxypropane |
b.  |
ii.1-Ethoxy- 2-nitrocyclohexane |
c.  |
iii. 4-Chloro-2,3-dimethylpentan-1-ol |
d.  |
iv. 2,6-Dimethylphenol |
The correct option is-
1. a=ii; b=iii; c=i; d=iv
2. a=iii; b=iv; c=i; d=ii
3. a=iv; b=iii; c=i; d=ii
4. a=iii; b=i; c=iv; d=ii

1. 2-Isopropyl-3-methyl-5-pentanol.
2. 3-Isopropyl-3-methyl-1-butanol.
3. 2,3,4-Trimethyl-1-pentanol.
4. 3,4,5-Trimethyl-1-hexanol.
\(CH_3−CH=CH_2 \xrightarrow{H_2O/H^+}\) A
| 1. | CH3CH2CH2OH | 2. | CH3CH(OH)CH3 |
| 3. | CH3CH(OH)CH2OH | 4. | CH3CH2CH3 |
Which of the following is an example of elimination reaction?
1. Chlorination of methane
2. Dehydration of ethanol
3. Nitration of benzene
4. Hydroxylation of ethylene
| Column A | Column B | ||
| i. | Lucas' reagent | a. | Alkaline KMnO4 |
| ii. | Baeyer's reagent | b. | CH3MgBr |
| iii. | PCC | c. | ZnCl2+HCl |
| iv. | Grignard reagent | d. |  |
Choose the correct answer from the options given below:
1. i‐c, ii‐d, iii‐a, iv‐b
2. i‐b, ii‐c, iii‐a, iv‐d
3. i‐b, ii‐d, iii‐a, iv‐c
4. i‐c, ii‐a, iii‐d, iv‐b
1. HCHO
2. CH3OH
3. CO2
4. CH3COCH3
What is the IUPAC name of the given compound?
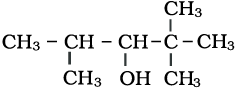
1. 2, 2, 2-Trimethylpentan-3-ol
2. 2, 2, 4-Trimethylpentan-3-ol
3. 2, 2, 3-Trimethylpentan-3-ol
4. 1, 1, 3-Trimethylpentan-3-ol
| Column I | Column II | ||
| Structural Formula | Compound Name | ||
| A. |  |
I. | Catechol |
| B. |  |
II. | Resorcinol |
| C. |  |
III. | Hydroquinone |
| D. |  |
IV. | o-Cresol |
| (A) | (B) | (C) | (D) | |
| 1. | (I) | (III) | (II) | (IV) |
| 2. | (III) | (I) | (II) | (IV) |
| 3. | (III) | (I) | (IV) | (II) |
| 4. | (I) | (III) | (IV) | (II) |











